近日,美国加州大学洛杉矶分校Keriann M. Backus团队报道了使用硅醚激活的化学蛋白质组学发现小分子结合位点。2026年4月27日,《自然-化学》杂志发表了这一成果。
在化学探针与药物发现研究中,基于质谱的化学蛋白质组学与光亲和标记相结合的方法已成为靶标发现和作用机制解析的主流策略。然而,光交联产生的肽段-化合物加合物为定量结合位点的发现带来了分析上的挑战。
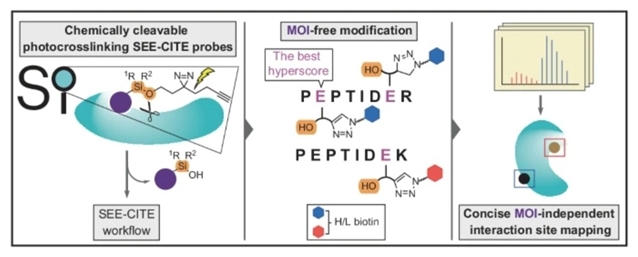
为应对这些难题,研究组建立了“硅醚赋能化学蛋白质组相互作用与靶标结合(SEE-CITE)”方法。SEE-CITE引入了一种全功能化的化学可裂解光交联手柄,能够实现标记位点的精准鉴定,并对不同化学结构化合物在结合位点上的相对占据程度进行头对头比较。
为确保标记残基的高置信度定位,他们对FragPipe计算平台中的MSFragger算法进行了扩展,使其能够针对光亲和标记和SEE-CITE数据输出定制化的定位得分。将该方法应用于筛选某些FDA批准的激酶抑制剂的先导片段及类似物时,SEE-CITE不仅解析了已知的药物结合位点,还发现了能够影响RTN4和COX5A蛋白活性的新型小分子结合位点。
附:英文原文
Title: Small-molecule binding-site discovery using silyl ether-enabled chemoproteomics
Author: Ngo, Chau, Takechi, Sho, Sivakumar, Aditya, Villanueva, Miranda, Yu, Fengchao, Ball, Andra B., Rubio, Javier, Biletch, Elijah, Burton, Nikolas R., Boatner, Lisa M., Kim, Phillip, Turmon, Alexandra C., Perumal, Nithesh, Liesa, Marc, Divakaruni, Ajit S., Nesvizhskii, Alexey I., Backus, Keriann M.
Issue&Volume: 2026-04-27
Abstract: For chemical probe and drug discovery campaigns, the pairing of mass spectrometry-based chemoproteomics with photoaffinity labelling has emerged as a favoured approach for target discovery and mode of action assignment. However, photocrosslinked peptide-compound adducts raise analytic challenges for quantitative binding site discovery. Here, to address these challenges, we establish the Silyl Ether Enables Chemoproteomic Interaction and Target Engagement (SEE-CITE) method. SEE-CITE incorporates a fully functionalized chemically cleavable photocrosslinking handle that enables precise site-of-labelling identification and head-to-head comparisons of relative binding site engagement by chemically diverse compounds. To ensure high-confidence localization of labelled residues, we extended the MSFragger algorithm of the FragPipe computational platform to report localization scores customized for photoaffinity labelling and SEE-CITE data. When applied to scout fragments and analogues of select FDA-approved kinase inhibitors, SEE-CITE delineates known drug binding sites and uncovers small-molecule binding sites that affect the protein activity of RTN4 and COX5A.
DOI: 10.1038/s41557-026-02127-4
Source: https://www.nature.com/articles/s41557-026-02127-4
Nature Chemistry:《自然—化学》,创刊于2009年。隶属于施普林格·自然出版集团,最新IF:24.274
官方网址:https://www.nature.com/nchem/
投稿链接:https://mts-nchem.nature.com/cgi-bin/main.plex
