近日,河南师范大学王键吉团队报道了高CuI含量及分子修饰铜基催化剂增强H2O活化的CO2选择性电合成乙醇。2026年3月30日,《美国化学会志》发表了这一成果。
利用二氧化碳和水电催化合成乙醇,是一条在产生增值化学品的同时闭合碳循环的有前景的策略。然而,在高电流密度下高选择性地生产乙醇仍然是一个巨大挑战。
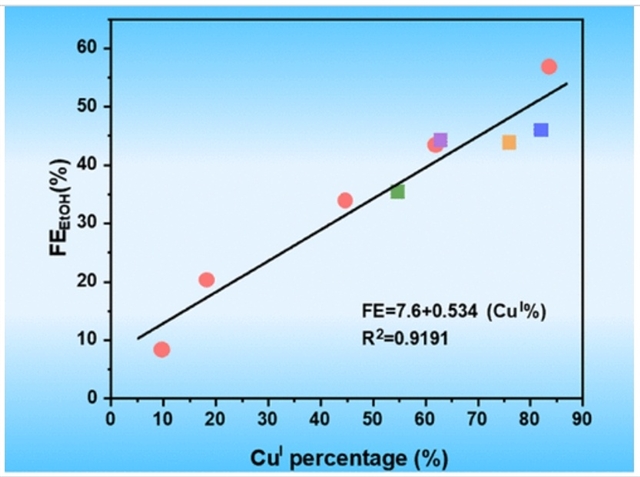
研究组通过在Cu2O表面覆盖一层配位络合物,设计了一系列铜基催化剂,以在CO2电还原过程中稳定Cu+。通过调控覆盖度,Cu+的原子百分比可从9.7%调节至83.6%;其对CO2电还原至乙醇的选择性从6.1%提升至56.8%,并且观察到Cu+原子百分比与乙醇选择性之间呈线性相关。值得注意的是,最优的(Cu+)83.6/Cu+BTEC催化剂实现了87.3%的C2+法拉第效率,部分电流密度达676.0 mA cm-2。
其中,乙醇的法拉第效率为56.8%,部分电流密度高达439.8 mA cm-2,接近先前报道的记录值,且该催化剂在110小时电解过程中保持稳定。原位光谱技术和DFT计算揭示,该催化剂降低了COatop–COH偶联的能垒,稳定了选择性决定中间体CH3CHO,并加速了水分子解离为活性氢(H),从而实现了优异的乙醇选择性生产。
附:英文原文
Title: Selective Electrosynthesis of Ethanol from CO2 Enabled by High CuI Content and Enhanced H2O Activation of Molecularly Modified Cu-Based Catalyst
Author: Nan Wang, Yuan Zhang, Zhaojun Min, Bing Chang, Huiyong Wang, Zhiyong Li, Maohong Fan, Suojiang Zhang, Jianji Wang
Issue&Volume: March 30, 2026
Abstract: Electrocatalytic synthesis of ethanol from CO2 and water is a promising strategy to close the carbon cycle while producing value-added chemicals. However, highly selective production of ethanol remains an enormous challenge especially at high current density. In this work, we design a series of copper (Cu)-based catalysts by covering a layer of coordination complex on the surface of Cu2O to stabilize CuI in the CO2 electroreduction process. By tuning the coverage, the atomic percent of CuI can be regulated from 9.7 to 83.6%, their selectivity for CO2 electroreduction to ethanol may be improved from 6.1 to 56.8%, and a linear correlation is observed between the CuI atomic percent and ethanol selectivity. Notably, the optimal (CuI)83.6/Cu+BTEC exhibits a C2+ Faradaic efficiency (FE) of 87.3% with a partial current density of 676.0 mA cm–2. In particular, the ethanol FE is 56.8%, and the partial current density is up to 439.8 mA cm–2, which is close to the record value reported previously, and the catalyst is stable in 110 h electrolysis. In situ spectroscopy techniques and DFT calculations reveal that the catalyst reduces the energy barrier of *COatop–*COH coupling, stabilizes the selectivity-determining intermediate CH3CHO*, and accelerates the dissociation of water molecules into active hydrogen (*H), thus resulting in an excellent selective production of ethanol.
DOI: 10.1021/jacs.5c19695
Source: https://pubs.acs.org/doi/abs/10.1021/jacs.5c19695
JACS:《美国化学会志》,创刊于1879年。隶属于美国化学会,最新IF:16.383
官方网址:https://pubs.acs.org/journal/jacsat
投稿链接:https://acsparagonplus.acs.org/psweb/loginForm?code=1000
