近日,苏州大学史海斌团队报道了光介导的抗原和核酸原位同步捕获可作为纳米疫苗促进肿瘤放射免疫治疗。该研究于2026年4月13日发表在《美国化学会志》上。
放射治疗诱导免疫原性癌细胞死亡以产生疫苗效应,是提高肿瘤治疗效果的一种有效策略。然而,该策略常常受到树突状细胞对释放的肿瘤抗原和损伤相关分子模式摄取不足的限制。
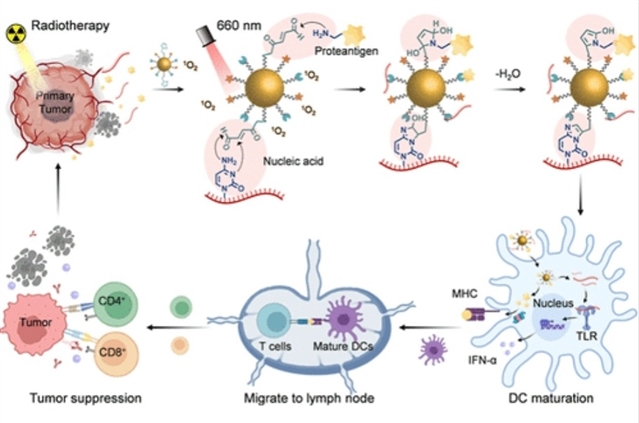
为解决这一问题,研究组报道了一种光介导的同步捕获抗原和核酸佐剂的策略,用于原位生成疫苗以增强肿瘤放射免疫治疗。通过在金纳米颗粒表面修饰3-(2-呋喃基)丙酸和光敏剂二氢卟吩e6,合理设计并制备了一种光响应纳米颗粒fAuNPs-Ce6。在660 nm激光照射下,Ce6产生的单线态氧催化下,fAuNPs-Ce6纳米颗粒能够通过呋喃介导的与氨基或碱基的共价反应,同时捕获肿瘤来源的蛋白抗原和核酸片段,从而显著延长抗原和佐剂在肿瘤微环境中的滞留时间,在不依赖外源性佐剂的情况下促进树突状细胞高效成熟,并引发强效的系统性抗肿瘤免疫。
更值得注意的是,这些纳米疫苗与PD-L1阻断联合使用,能够显著增强放射治疗效果,实现对原发灶和远端肿瘤生长的显著抑制。因此,这种光介导的原位疫苗接种策略为恶性肿瘤的精准治疗提供了一种有前景的方法。
附:英文原文
Title: Light-Mediated In Situ Synchronous Capture of Antigens and Nucleic Acids as Nanovaccines to Boost Tumor Radioimmunotherapy
Author: Miao Li, Jinfeng Zhu, Zhengzhong Lv, Zhixin Han, Yuqi Zhang, Xingxiang Ren, Yurong Fan, Hongyuan Wen, Yiming Feng, Yan Chen, Yongxiang Bai, Danyang Ji, Xiaju Cheng, Haibin Shi
Issue&Volume: April 13, 2026
Abstract: Radiotherapy-induced immunogenic cancer cell death to generate vaccine effects is an effective approach to enhancing the efficacy of tumor therapy. However, it is often constrained by insufficient uptake of released tumor antigens and damage-associated molecular patterns (DAMPs) by dendritic cells (DCs). To address this issue, a light-mediated strategy for synchronous capture of antigens and nucleic acid adjuvants is reported to in situ generate vaccines for enhanced tumor radioimmunotherapy. A type of light-responsive nanoparticle fAuNPs-Ce6 is rationally designed and fabricated by modifying 3-(2-furyl) propanoic acids and photosensitizer Chlorin e6 (Ce6) on the surface of gold nanoparticles. Under the catalysis of 1O2 generated from Ce6 upon a 660 nm laser irradiation, fAuNPs-Ce6 nanoparticles can capture the tumor-derived protein antigens and nucleic acid fragments simultaneously through the furan-mediated covalent reactions with amino group or nucleobase, which significantly prolongs the retention time of antigens and adjuvants in the tumor microenvironment, promoting robust DC maturation without exogenous adjuvants and eliciting potent systemic antitumor immunity. More notably, these nanovaccines in combination with PD-L1 blockade can remarkably enhance the radiotherapeutic efficacy, achieving significant suppression of both primary and distant tumor growth. Therefore, this light-mediated in situ vaccination may offer a promising strategy for the precise treatment of malignant tumors.
DOI: 10.1021/jacs.5c21345
Source: https://pubs.acs.org/doi/abs/10.1021/jacs.5c21345
JACS:《美国化学会志》,创刊于1879年。隶属于美国化学会,最新IF:16.383
官方网址:https://pubs.acs.org/journal/jacsat
投稿链接:https://acsparagonplus.acs.org/psweb/loginForm?code=1000
