SCFFBXO42的模板驱动支架调控PP2A的降解,这一成果由发现肿瘤科Robert L. Yauch团队经过不懈努力而取得。这一研究成果发表在2026年4月15日出版的国际学术期刊《自然》上。
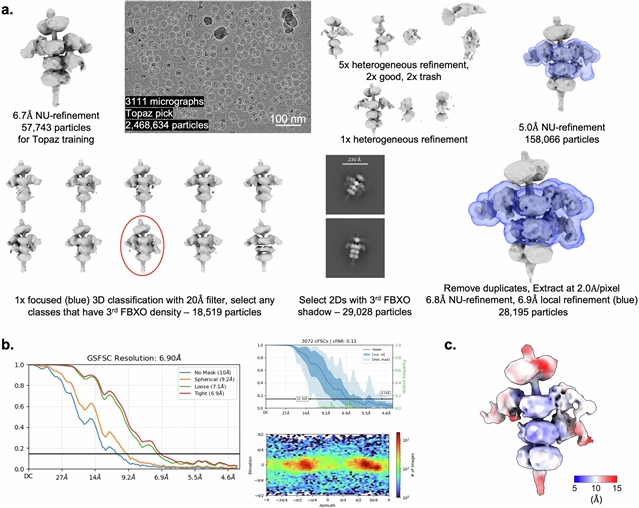
本研究发现E3连接酶SKP1-CUL1-F-box (SCF)复合物含有F-box其他蛋白42 (FBXO42,也称为JFK;以下简称SCFFBXO42)在与螺旋状蛋白CCDC6的复合物中降解无全酶PP2Ac,以维持癌细胞的适应性。FBXO42 - CCDC6 - PP2Ac组装体的低温电镜结构揭示了假对称结构,其中CCDC6形成中心二聚体模板,招募PP2Ac的多个拷贝并为FBXO42创建底物。CCDC6-PP2Ac异源二聚体的四元结构和PP2Ac翻译后甲基化的尾部都被FBXO42识别为泛素化。CCDC6促进的多价结构使多个降解复合物沿着单个线圈组装,导致游离磷酸酶的周转和催化活性的下调。总之,他们的发现定义了一种通过泛素-蛋白体系统控制PP2A的机制,并建立了cullin-RING连接酶-底物相互作用的范例。
据介绍,蛋白磷酸酶2A (PP2A)是一种丝氨酸/苏氨酸磷酸酶,它调节几乎所有细胞过程的磷酸化,包括细胞分裂和增殖。PP2A形成异三聚体全酶复合物,包括催化亚基(PP2Ac)、脚手架亚基(PP2Aa)和可变B调节亚基,这些亚基精确控制酶底物特异性,防止磷酸化蛋白的不加区分地去磷酸化。然而,控制非络合催化亚基活性的机制仍然相对不清楚。
附:英文原文
Title: Template-driven scaffolding of SCFFBXO42 regulates PP2A degradation
Author: Coassolo, Sebastien, Michaelian, Nairie, Maculins, Timurs, Azumaya, Caleigh M., Cheung, Tommy K., Yin, Jianping, Zilberleyb, Inna, Pahuja, Kanika Bajaj, Garner, Thomas, Lau, Ted, Mau, Davis, Grimmer, Matthew, Fortin, Jean-Philippe, Costa, Mike, Dimitrova, Yoana N., Rose, Christopher M., Hsu, Peter L., Yauch, Robert L.
Issue&Volume: 2026-04-15
Abstract: Protein phosphatase 2A (PP2A) is a Ser/Thr phosphatase that regulates the phosphorylation of almost all cellular processes, including cell division and proliferation1,2. PP2A forms heterotrimeric holoenzyme complexes comprising a catalytic subunit (PP2Ac), a scaffolding subunit (PP2Aa) and variable B regulatory subunits that exert precise control over enzyme substrate specificity and prevent indiscriminate dephosphorylation of phosphoproteins3. However, the mechanisms that control the activity of uncomplexed catalytic subunits have remained relatively unclear. Here we find that the E3 ligase SKP1–CUL1–F-box (SCF) complex containing F-box other protein 42 (FBXO42, also known as JFK; hereafter, SCFFBXO42) degrades holoenzyme-free PP2Ac in a complex with the coiled-coil protein CCDC6 to maintain cancer cell fitness. The cryo-electron microscopy structure of the FBXO42–CCDC6–PP2Ac assembly reveals a pseudosymmetric architecture in which CCDC6 forms a central dimeric template that recruits multiple copies of PP2Ac and creates a substrate for FBXO42. Both the quaternary structure of this CCDC6–PP2Ac heterodimer and the post-translationally methylated tail of PP2Ac are recognized by FBXO42 for ubiquitination. The multivalent structure facilitated by CCDC6 enables the assembly of multiple degradation complexes along a single coiled coil, leading to the turnover of free phosphatases and downregulation of catalytic activity. Together, our findings define a mechanism for PP2A control through the ubiquitin-proteosome system and establish a paradigm for cullin-RING ligase–substrate interactions.
DOI: 10.1038/s41586-026-10368-z
Source: https://www.nature.com/articles/s41586-026-10368-z
Nature:《自然》,创刊于1869年。隶属于施普林格·自然出版集团,最新IF:69.504
官方网址:http://www.nature.com/
投稿链接:http://www.nature.com/authors/submit_manuscript.html
