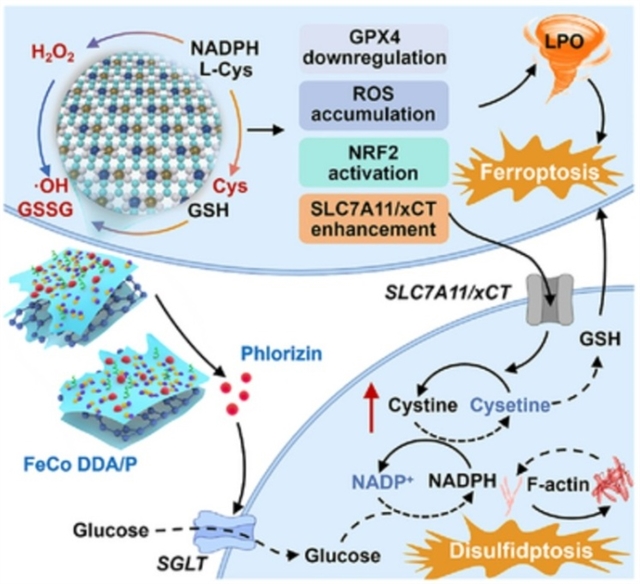
近日,中国科学院长春应用化学研究所张洪杰团队报道了电荷可调致密双原子纳米酶通过多酶模拟催化重编程生物硫醇代谢,协同诱导铁死亡和二硫死亡。这一研究成果于2026年4月6日发表在《德国应用化学》杂志上。
生物硫醇在维持细胞氧化还原稳态、协调程序性细胞死亡通路以及调节免疫反应中发挥着关键作用。因此,重编程肿瘤细胞中的生物硫醇代谢,为增强抗肿瘤免疫提供了一种有前景的策略。
研究组报告了一种合理设计的、高密度可及异核Fe/Co双原子纳米酶(FeCo DDA)。该纳米酶通过配位高密度的Fe和Co活性中心来调控生物硫醇代谢并诱导细胞内二硫化物积累,从而模拟天然黄素酶。致密的异核双原子催化中心(Fe和Co的质量分数分别为10.35%和11.32%)通过调节电子重新分布,优化了Bader电荷和d带中心,使其具有优异的模拟酶活性,能够催化生物硫醇的氧化,破坏肿瘤细胞的稳态。同时,共负载的根皮苷抑制葡萄糖摄取,进一步驱动补偿性胱氨酸积累并破坏谷胱甘肽的生物合成。
这种双重作用协同诱导铁死亡增强的二硫死亡,破坏氧化还原稳态并触发免疫原性细胞死亡。因此,共负载根皮苷的FeCo DDA(FeCo DDA/P)不仅能增强肿瘤细胞的免疫原性,还能重塑免疫抑制性肿瘤微环境,从而增强抗肿瘤免疫反应。这项工作突出了利用双原子纳米酶策略重编程肿瘤代谢并协同多模式细胞死亡,以实现有效的肿瘤免疫治疗。
附:英文原文
Title: Charge-Tunable Dense Dual-Atom Nanozymes Reprogram Biothiol Metabolism Through Multi-Enzyme-Mimetic Catalysis to Synergistically Induce Ferroptosis and Disulfidptosis
Author: Rui Niu, Yang Liu, Bin Zhang, Bo Xu, Zhijia Lv, Ruiping Deng, Shuyan Song, Kai Liu, Yinghui Wang, Hongjie Zhang
Issue&Volume: 2026-04-06
Abstract: Biothiols play a pivotal role in maintaining cellular redox homeostasis, coordinating programmed cell death pathways, and modulating immune responses. Reprogramming biothiol metabolism in tumor cells thus presents a promising strategy for enhancing anti-tumor immunity. Here, we report the rational design of a densely accessible heteronuclear Fe/Co dual-atom nanozyme (FeCo DDA), which mimics natural flavoenzymes by coordinating high-density Fe and Co active centers to regulate biothiol metabolism and induce intracellular disulfide accumulation. The dense heteronuclear diatomic catalytic center (with Fe and Co mass fractions of 10.35% and 11.32%, respectively) optimizes the Bader charge and d-band center by adjusting electron redistribution, endowing it with excellent mimetic enzymatic activities for catalyzing the oxidation of biothiols to disrupt the homeostasis of tumor cells. Simultaneously, co-loaded phlorizin inhibits glucose uptake, further driving compensatory cystine accumulation and disrupting glutathione biosynthesis. This dual action synergistically induces ferroptosis-enhanced disulfidptosis, disrupting redox homeostasis and triggering immunogenic cell death. As a result, FeCo DDA co-loaded with phlorizin (FeCo DDA/P) not only enhances tumor cell immunogenicity but also reshapes the immunosuppressive tumor microenvironment, thereby potentiating anti-tumor immune responses. This work highlights a dual-atom nanozyme strategy to reprogram tumor metabolism and orchestrate multimodal cell death for effective tumor immunotherapy.
DOI: 10.1002/anie.202524634
Source: https://onlinelibrary.wiley.com/doi/10.1002/anie.202524634
Angewandte Chemie:《德国应用化学》,创刊于1887年。隶属于德国化学会,最新IF:16.823
官方网址:https://onlinelibrary.wiley.com/journal/15213773
投稿链接:https://www.editorialmanager.com/anie/default.aspx
