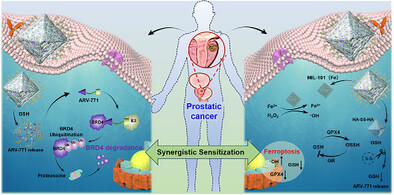
近日,武汉大学李伟团队报道了纳米酶介导的PROTACs递送用于前列腺癌的靶向蛋白降解和铁死亡增敏。这一研究成果于2026年3月23日发表在《德国应用化学》杂志上。
去势抵抗性前列腺癌(CRPC)因对传统雄激素受体(AR)靶向疗法具有耐药性,仍是临床面临的一大挑战。蛋白降解靶向嵌合体(PROTAC)药物(如ARV-771)虽能选择性降解致癌驱动蛋白,但其递送难题严重制约了疗效与安全性。
研究组报道了一种纳米工程化PROTAC平台——ARV@MIL-HA-ss-HA,该平台显著提升了ARV-771的递送效率、药代动力学表现及治疗效果。该系统采用MIL-101纳米颗粒作为载体兼纳米酶,并通过透明质酸-二硫键-透明质酸(HA-ss-HA)水凝胶修饰,实现CD44介导的肿瘤靶向及谷胱甘肽(GSH)触发的药物释放。借助MIL-101的催化活性与HA-ss-HA的GSH耗竭能力,ARV@MIL-HA-ss-HA可将细胞内H2O2转化为羟基自由基(·OH)并抑制GSH水平,从而诱导铁死亡。
与此同时,ARV-771介导的BRD4降解使肿瘤细胞对铁死亡增敏,形成双重作用的协同机制。在CRPC模型中的体外与体内研究证实,该平台可实现高效的BRD4降解、增强的铁死亡细胞死亡及卓越的抗肿瘤疗效,且全身毒性极低。该研究成果将这种纳米PROTAC策略定位为一种具有临床转化前景的双机制疗法,有望克服CRPC的耐药性难题。
附:英文原文
Title: Nanozyme-Mediated PROTACs Delivery for Targeted Protein Degradation and Ferroptosis Sensitization in Prostate Cancer
Author: Chenyuan Wang, Xue Jiang, Jiapeng Lei, Wen Zhang, Fuqiang Shao, Tianshu Hao, Yongnian Zeng, Peng Jiang, Fan Cheng, Lingqi Liu, Wei Li
Issue&Volume: 2026-03-23
Abstract: Castration-resistant prostate cancer (CRPC) remains a major clinical challenge due to its resistance to conventional androgen receptor (AR)-targeted therapies. Proteolysis-targeting chimeras (PROTACs) drugs, such as ARV-771, can selectively degrade cancer-driving proteins but face major delivery challenges that limit their efficacy and safety. Here, we report a nanoengineered PROTAC platform, ARV@MIL-HA-ss-HA, that markedly improves ARV-771 delivery, pharmacokinetics, and therapeutic efficacy. The system employs MIL-101 nanoparticles as both a carrier and a nanozyme, modified with hyaluronic acid-disulfide-hyaluronic acid (HA-ss-HA) hydrogel to achieve CD44-mediated tumor targeting and glutathione (GSH)-triggered release. Leveraging the catalytic activity of MIL-101 and the GSH-depleting capacity of HA-ss-HA, ARV@MIL-HA-ss-HA converts intracellular H2O2 into hydroxyl radicals (·OH) and suppresses GSH levels, thereby inducing ferroptosis. Concurrently, ARV-771-mediated BRD4 degradation sensitizes tumor cells to ferroptosis, establishing a dual-action synergistic mechanism. In vitro and in vivo studies in CRPC models confirmed efficient BRD4 degradation, enhanced ferroptotic cell death, and superior antitumor efficacy with minimal systemic toxicity. Our findings position this nano-PROTAC strategy as a clinically promising dual-mechanism therapy capable of overcoming resistance in CRPC.
DOI: 10.1002/anie.6657773
Source: https://onlinelibrary.wiley.com/doi/10.1002/anie.6657773
Angewandte Chemie:《德国应用化学》,创刊于1887年。隶属于德国化学会,最新IF:16.823
官方网址:https://onlinelibrary.wiley.com/journal/15213773
投稿链接:https://www.editorialmanager.com/anie/default.aspx
