近日,英国剑桥大学Balasubramanian, Shankar团队报道了利用基于G4配体的靶向蛋白水解嵌合体降解染色质中的G-四联体结合蛋白。这一研究成果发表在2026年3月19日出版的《自然-化学》杂志上。
靶向蛋白降解技术能够干预疾病相关蛋白的功能,但现有方法大多依赖于蛋白靶点的直接配体结合,这限制了其在难以实现选择性结合的蛋白质中的应用。
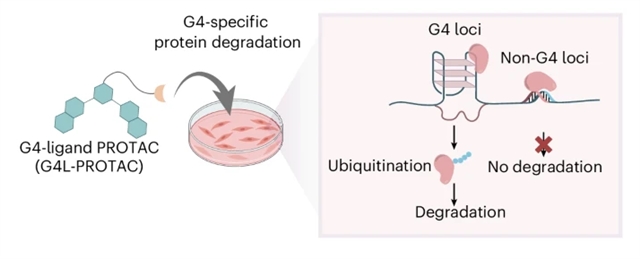
研究组提出了一种概念新颖的策略,可在染色质环境中降解与DNA G-四链体二级结构相关的蛋白质。G-四链体是一种非经典核酸结构,在开放染色质中形成于转录活跃基因的调控区域,在癌症状态下尤为丰富。虽然众多蛋白质能够识别或调控G-四链体结构,但在天然染色质环境中选择性靶向G-四链体结合蛋白仍具挑战。
研究组设计的双功能分子是一种靶向嵌合体,可结合天然存在的G-四链体,招募E3泛素连接酶,进而降解G-四链体特异性转录因子及染色质重塑因子(如FUS、SMARCA4和ATRX)。这些蛋白在转录调控和DNA修复中发挥关键作用,是重要的治疗靶点。该研究提出的策略有望发展为靶向治疗以G-四链体异常活性为特征的疾病(如癌症)的新方法。
附:英文原文
Title: Degradation of G-quadruplex-binding proteins in chromatin using G4-ligand-based proteolysis-targeting chimeras
Author: Wang, Zixuan, He, Xuan, Zhang, Xiaoyun, Spiegel, Jochen, Flynn, Sean M., Balasubramanian, Shankar
Issue&Volume: 2026-03-19
Abstract: Targeted protein degradation can intervene with the function of disease-related proteins, but most current approaches rely on direct ligand engagement of a protein target, limiting their applicability to proteins that are difficult to bind selectively. Here we present a conceptually unique approach to degrade proteins associated with DNA G-quadruplex (G4) secondary structures in a chromatin context. G4s are non-canonical nucleic acid structures that form at regulatory regions of transcriptionally active genes in open chromatin, and are abundant in cancer states. Although many proteins recognize or regulate G4 structures, selectively targeting G4-binding proteins in their native chromatin environment is challenging. Our bifunctional molecules are proteolysis-targeting chimeras that bind naturally occurring G4s, recruit E3 ubiquitin ligases and degrade G4-specific transcription factors and chromatin remodellers such as FUS, SMARCA4 and ATRX. These proteins are important therapeutic targets that play crucial roles in transcription regulation and DNA repair. Our approach has the potential to be exploited in a therapeutic strategy to target diseases characterized by aberrant G4 activity, such as cancers.
DOI: 10.1038/s41557-026-02111-y
Source: https://www.nature.com/articles/s41557-026-02111-y
Nature Chemistry:《自然—化学》,创刊于2009年。隶属于施普林格·自然出版集团,最新IF:24.274
官方网址:https://www.nature.com/nchem/
投稿链接:https://mts-nchem.nature.com/cgi-bin/main.plex
