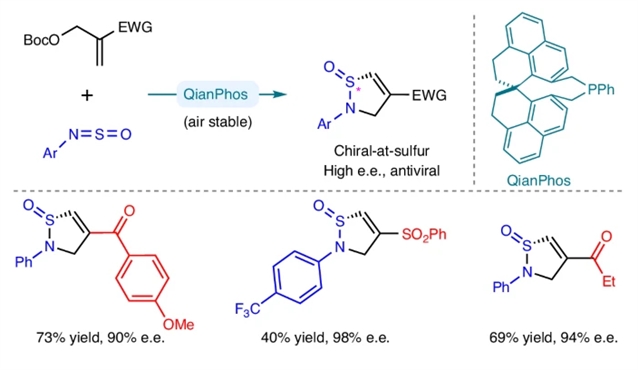
近日,香港科技大学孙建伟团队报道了空气稳定手性膦催化S(IV)-立体亚胺的有机催化对映选择性合成。相关论文于2026年3月10日发表在《自然-化学》杂志上。
具有硫手性的富集对映体乙烯基亚磺酰胺在药物化学中具有重要价值,但传统合成策略难以实现其高效制备。
研究组报道了一种有机催化新方法,通过设计的手性有机膦催化剂,实现了Morita-Baylis-Hillman酯与亚磺酰亚胺之间的对映选择性C-S键构建。该催化剂的结构刚性不仅确保了反应优异的化学选择性、对映选择性和非对映选择性,还赋予其卓越的空气稳定性。
密度泛函理论计算与实验研究表明,鏻物种可能是催化剂的休息态,而亚磺酰亚胺可能同时作为反应底物和促进剂参与关键鏻中间体的形成。环状乙烯基亚磺酰胺对严重急性呼吸综合征冠状病毒2突变型刺突蛋白及人类免疫缺陷病毒1型包膜蛋白表现出良好的结合亲和力,表明这一尚待深入探索的化学空间在抗病毒药物研发中具有巨大潜力。
附:英文原文
Title: Organocatalytic enantioselective synthesis of S(IV)-stereogenic sulfinamides enabled by an air-stable chiral phosphine
Author: Qian, Chenxiao, Chen, Yuyang, Wang, Baocheng, Wang, Tao, Zhang, Chaoshen, Lin, Zhenyang, Sun, Jianwei
Issue&Volume: 2026-03-10
Abstract: Enantioenriched vinyl sulfinamides with chiral-at-sulfur chirality are medicinally valuable but have limited accessibility with conventional strategies. Here we disclose an organocatalytic approach via enantioselective CS bond formation between Morita–Baylis–Hillman esters and sulfinylamines catalysed by a designed chiral organophosphine. The structural rigidity of this catalyst is crucial for not only the excellent chemo-, enantio- and diastereoselectivities in this process but also its extraordinary air stability. Density functional theory and experimental studies indicated that the phosphonium species probably serves as the catalyst resting state, and the sulfinylamine may play a dual role as both reaction partner and promoter for the formation of the key phosphonium intermediate. The cyclic vinyl sulfinamides showed promising binding affinity to the mutant spike of severe acute respiratory syndrome coronavirus 2 and ENV of human immunodeficiency virus-1, suggesting that this less-explored chemical space has great potential for further antiviral drug development.
DOI: 10.1038/s41557-026-02095-9
Source: https://www.nature.com/articles/s41557-026-02095-9
Nature Chemistry:《自然—化学》,创刊于2009年。隶属于施普林格·自然出版集团,最新IF:24.274
官方网址:https://www.nature.com/nchem/
投稿链接:https://mts-nchem.nature.com/cgi-bin/main.plex
