近日,西北大学魏颢团队研究了单硅插入对苯并呋喃扩环的影响。相关论文于2026年3月9日发表在《美国化学会志》上。
鉴于有机硅化合物在药物化学中的重要性,将硅原子插入易得杂环是一种强大但尚未充分发展的骨架跳跃策略,可用于扩展类药化学空间。
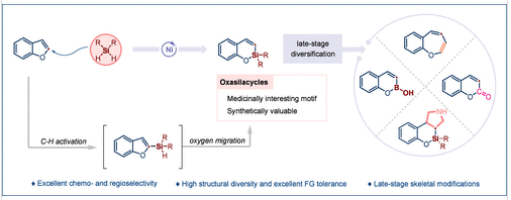
研究组报道了镍催化的单硅原子插入反应,实现了将易得的苯并呋喃转化为相应的高价值含氧硅环。该反应涉及苯并呋喃的C-H键硅基化和1,2-氧迁移过程。所得到的含氧硅环可作为通用合成子,用于生成多种化合物,并可获得不同的环状骨架,如氧杂硼杂环、香豆素类、苯并氧杂环庚烷和多环氧杂硅烷,从而使初始苯并呋喃核心多样化。该研究为苯并呋喃的骨架编辑建立了一个多功能平台,拓宽了药物化学的合成工具箱。
附:英文原文
Title: Ring Expansion of Benzofurans by Single-Silicon Insertion
Author: Qiuling Yan, Hongxia Song, Hong Lu, Hao Wei
Issue&Volume: March 9, 2026
Abstract: Given the importance of organosilicon compounds in medicinal chemistry, the insertion of a silicon atom into a readily available heterocycle is a powerful yet underdeveloped scaffold-hopping strategy for expanding drug-like chemical spaces. Herein, we report a Ni-catalyzed single-silicon atom insertion reaction for the transformation of readily available benzofurans into their corresponding high-value oxasilacycles. The reaction involves the C–H bond silylation of benzofurans and 1,2-oxygen migration. The resulting oxasilacycles serve as versatile synthons for generating various compounds and provide access to different cyclic skeletons such as oxaborins, coumarins, benzooxepines, and polycyclic oxasilaalkanes, thereby diversifying the initial benzofuran core. This study establishes a versatile platform for the skeletal editing of benzofurans, broadening the synthetic toolbox for medicinal chemistry.
DOI: 10.1021/jacs.6c00274
Source: https://pubs.acs.org/doi/abs/10.1021/jacs.6c00274
JACS:《美国化学会志》,创刊于1879年。隶属于美国化学会,最新IF:16.383
官方网址:https://pubs.acs.org/journal/jacsat
投稿链接:https://acsparagonplus.acs.org/psweb/loginForm?code=1000
