哺乳动物昼夜转换的细胞基础,这一成果由英国剑桥MRC分子生物学实验室
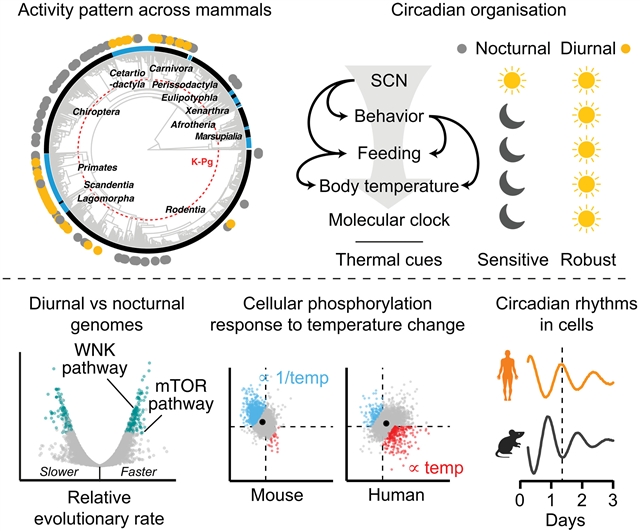
研究人员发现了一个保守的细胞固有的热力学机制,可能促进了这种转变。在昼行性哺乳动物的细胞中,蛋白质合成、磷酸化和昼夜节律对温度变化的敏感性低于夜行性哺乳动物的细胞。比较基因组学揭示了基本信号通路的加速进化,包括雷帕霉素(mTOR)的机制靶点,增加了细胞昼夜时钟对热和渗透扰动的灵活性。在夜间活动的小鼠中,mTOR抑制使细胞、组织和行为转向昼间活动。这些发现揭示了昼夜转换的遗传和生化基础,强调了细胞信号网络如何编码复杂的表型,如时间生态位选择。
研究人员表示,早期哺乳动物在夜间活动,而恐龙在白天活动。白垩纪-古近纪灭绝后,哺乳动物向日间活动的转变加速,但潜在的机制尚不清楚。
附:英文原文
Title: A cellular basis for the mammalian nocturnal-diurnal switch
Author: Andrew D. Beale, Matthew J. Christmas, Nina M. Rzechorzek, Andrei Mihut, Aiwei Zeng, Christopher Ellis, Nathan R. James, Nicola J. Smyllie, Violetta Pilorz, Rose Richardson, Mads F. Bertelsen, Shaline V. Fazal, Zanna Voysey, Kevin Moreau, Jerry Pelletier, Priya Crosby, Sew Y. Peak-Chew, Rachel S. Edgar, Madeline A. Lancaster, Roelof A. Hut, John S. O’Neill
Issue&Volume: 2026-02-26
Abstract: Early mammals were nocturnal while dinosaurs dominated the daytime. Mammalian transition to daytime activity accelerated after the Cretaceous-Paleogene extinction, but the underlying mechanisms remain unclear. We identified a conserved cell-intrinsic, thermodynamic mechanism that likely facilitated this shift. In cells from diurnal mammals, protein synthesis, phosphorylation, and circadian timing were less sensitive to temperature changes than were cells from nocturnal mammals. Comparative genomics revealed accelerated evolution within essential signaling pathways, including mechanistic target of rapamycin (mTOR), that increase the robustness of diurnal cellular clocks to thermal and osmotic perturbation. In nocturnal mice, mTOR inhibition shifted cells, tissues, and behavior toward diurnal activity. These findings uncover a genetic and biochemical basis for nocturnal-diurnal switching, emphasizing how cellular signaling networks can encode complex phenotypes such as temporal niche selection.
DOI: ady2822
Source: https://www.science.org/doi/10.1126/science.ady2822
