近日,英国牛津大学Edward A. Anderson团队研究了杂[3.1.1]螺桨烷。该成果于2026年2月25日发表在《自然-化学》杂志上。
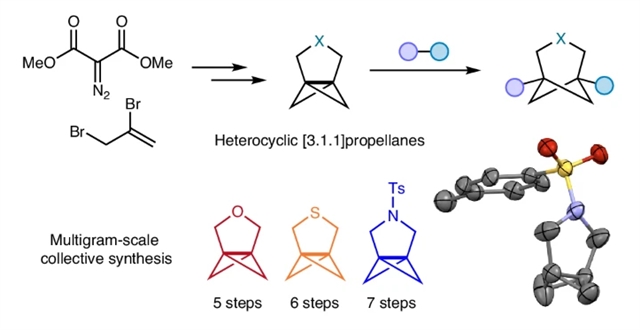
[n.1.1]螺桨烷是双环[n.1.1]烷烃的关键前体,这类刚性小环烃类化合物作为当代药物设计中二取代苯环的生物电子等排体,已成为重要的结构单元。含杂环的[n.1.1]螺桨烷可直接合成结构多样化的桥联双环杂环化合物,相较于已建立的碳环类似物,这些杂环应具有更优异的理化性质。
研究组报道了在三元桥上含有氧、氮、硫杂原子的杂环[3.1.1]螺桨烷家族的统一合成方法。该发展合成路径必然不同于已建立的碳环螺桨烷合成路线,采用通过铑催化环丙烷化反应便捷构建的克级规模通用前体。这些杂[3.1.1]螺桨烷可发生多种自由基开环反应,获得在新药研发项目中具有高度实用价值的桥联杂环化合物。
附:英文原文
Title: Hetero[3.1.1]propellanes
Author: Revie, Rebecca I., Dasgupta, Ayan, Biddick, Yasmine, Christensen, Kirsten E., Smith, Russell C., Anderson, Edward A.
Issue&Volume: 2026-02-25
Abstract: [n.1.1]Propellanes are key precursors to bicyclo[n.1.1]alkanes, rigid small-ring hydrocarbons that have emerged as important building blocks in contemporary drug design as bioisosteres for disubstituted benzene rings. [n.1.1]Propellanes featuring heterocyclic rings could enable the direct synthesis of a wide diversity of bridged bicyclic heterocycles, which should exhibit superior physicochemical profiles compared to their established carbocyclic analogues. Here we report the unified synthesis of a family of heterocyclic [3.1.1]propellanes featuring oxygen, nitrogen and sulfur heteroatoms in the three-carbon bridge. The approaches we have developed are necessarily distinct from the established routes to carbocyclic propellanes, and utilize a common precursor that is conveniently assembled on a multigram scale via rhodium-catalysed cyclopropanation. These hetero[3.1.1]propellanes undergo a range of radical ring-opening reactions, affording bridged heterocycles that are of high utility in drug-discovery programmes.
DOI: 10.1038/s41557-026-02072-2
Source: https://www.nature.com/articles/s41557-026-02072-2
Nature Chemistry:《自然—化学》,创刊于2009年。隶属于施普林格·自然出版集团,最新IF:24.274
官方网址:https://www.nature.com/nchem/
投稿链接:https://mts-nchem.nature.com/cgi-bin/main.plex
