近日,南开大学朱守非团队研究了末端炔烃铁催化反马尔可夫尼科夫烯丙基锌化。2026年2月25日出版的《美国化学会志》发表了这项成果。
末端炔烃的烯丙基金属化反应主要取决于底物的电子性质,因此对于普通末端炔烃的反马氏加成(烯丙基加成到末端碳原子)缺乏有效策略。
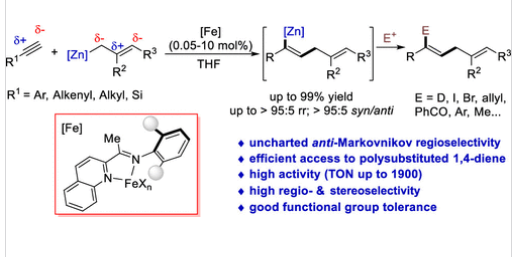
研究组报道了2-(亚胺)喹啉铁催化的芳基/烷基取代末端炔烃反马氏烯丙基锌化反应。该方案以高达99%的收率获得反马氏加成产物1,4-二烯基锌试剂,具有优异的区域选择性和立体选择性(多数>95:5),底物适用范围广且官能团兼容性好。该反应为现有方法无法获得的1,4-二烯基锌试剂提供了直接高效的合成途径,并成功应用于多取代1,4-二烯及多种天然产物关键中间体的简洁合成。
机理研究表明,Fe(0)催化剂对炔烃的活化作用与2-(亚胺)喹啉配体的半开放空间位阻效应是克服电子效应偏向、实现目标选择性的关键。该工作凸显了低价态铁催化剂在调控经典有机反应选择性方面的独特优势,为新型1,4-二烯化合物的合成提供了有效方法。
附:英文原文
Title: Iron-Catalyzed anti-Markovnikov Allylzincation of Terminal Alkynes
Author: Lu-Jie Li, Peng He, Xin-Yu Zhang, Shou-Fei Zhu
Issue&Volume: February 25, 2026
Abstract: Allylmetalation of terminal alkynes is predominantly governed by substrate electronic properties, leaving no effective strategy for anti-Markovnikov addition (allyl addition to the terminal carbon) of general terminal alkynes. Herein, we report a 2-(imino)quinoline iron-catalyzed anti-Markovnikov allylzincation of aryl- and alkyl-substituted terminal alkynes. This protocol affords anti-Markovnikov addition products, 1,4-dienylzinc reagents, in high yields (up to 99%) with excellent regio- and stereoselectivities (mostly >95:5), featuring broad substrate scope and good functional group tolerance. It provides a direct, efficient route to 1,4-dienylzinc reagents which are inaccessible via reported methods, and has been applied to the concise synthesis of polysubstituted 1,4-dienes, as well as key intermediates for several natural products. Mechanistic studies reveal that alkyne activation by Fe(0) catalyst and the semiopen steric effect of 2-(imino)quinoline ligands are critical to overcoming electronic biases and achieving the desired selectivity. This work highlights the unique advantages of low-valent iron catalysts in regulating classical organic reaction selectivity and offers an effective method for accessing novel 1,4-dienes.
DOI: 10.1021/jacs.5c21100
Source: https://pubs.acs.org/doi/abs/10.1021/jacs.5c21100
JACS:《美国化学会志》,创刊于1879年。隶属于美国化学会,最新IF:16.383
官方网址:https://pubs.acs.org/journal/jacsat
投稿链接:https://acsparagonplus.acs.org/psweb/loginForm?code=1000
