湖北大学李壮团队的一项最新研究报道了靶DNA诱导丝的形成和SPARDA复合体的核酸酶激活。这一研究成果发表在2025年3月24日出版的国际学术期刊《细胞研究》上。
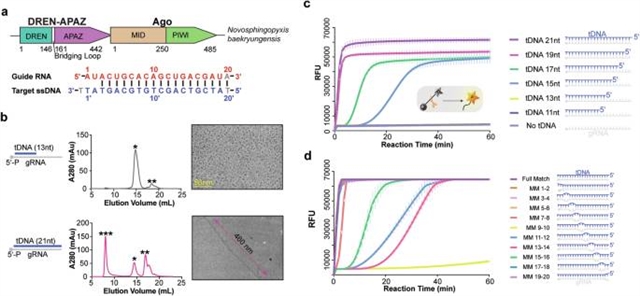
在这项研究中,课题组研究人员研究了Novosphingopyxis baekryungensis (NbaSPARDA)的SPARDA系统,并发现了一个意想不到的丝结构,在目标DNA结合时,它与侧支核酸酶活性密切相关。丝的形成和核酸酶的激活需要一个长度足够且在中心区域具有完美互补的引导靶异双工。以约3.0的分辨率测定了负载引导RNA、不同长度的靶DNA和底物ssDNA的NbaSPARDA复合物的一系列低温EM结构。结构分析表明,引导RNA结合诱导NbaSPARDA复合体的二聚化,而靶DNA结合破坏这种二聚化。导靶异质双工的进一步传播通过检查点机制触发细丝的形成。NbaSPARDA丝由一个由连锁短Argonaute蛋白组成的骨架组成,内层由DREN核酸酶结构域组成。细丝的形成导致单体DREN核酸酶结构域的四聚化,激活其对环境核酸的附带核酸酶活性-这是分子诊断的一个特征。对于异源表达NbaSPARDA系统的细菌,防御入侵的噬菌体和质粒依赖于丝的形成。总的来说,这些发现阐明了NbaSPARDA复合体的详细工作机制,并强调了其丝形成在宿主防御中的重要性。
据了解,基于短Argonaute的细菌防御系统SPARDA (short Prokaryotic Argonaute and DNase/RNase-APAZ)利用引导RNA靶向入侵的互补DNA,并表现出附带的核酸酶活性,导致细胞死亡或休眠。然而,其详细机制仍然知之甚少。
附:英文原文
Title: Target DNA-induced filament formation and nuclease activation of SPARDA complex
Author: Wang, Feng, Xu, Haijiang, Zhang, Chendi, Xue, Jialin, Li, Zhuang
Issue&Volume: 2025-03-24
Abstract: The short Argonaute-based bacterial defense system, SPARDA (Short Prokaryotic Argonaute and DNase/RNase-APAZ), utilizes guide RNA to target invading complementary DNA and exhibits collateral nuclease activity, leading to cell death or dormancy. However, its detailed mechanisms remain poorly understood. In this study, we investigated the SPARDA system from Novosphingopyxis baekryungensis (NbaSPARDA) and discovered an unexpected filament configuration upon target DNA binding, which strongly correlated with collateral nuclease activity. Filament formation and nuclease activation require a guide–target heteroduplex of sufficient length with perfect complementarity at the central region. A series of cryo-EM structures of NbaSPARDA complexes, loaded with guide RNA, target DNA of varying lengths, and substrate ssDNA, were determined at ~3.0 resolution. Structural analyses indicated that guide RNA binding induces dimerization of the NbaSPARDA complex, while target DNA engagement disrupts this dimerization. Further propagation of the guide–target heteroduplex triggers filament formation through a checkpoint mechanism. The NbaSPARDA filament consists of a backbone formed by interlocking short Argonaute proteins, with an inner layer composed of DREN nuclease domains. Filament formation leads to tetramerization of the monomeric DREN nuclease domain, activating its collateral nuclease activity against environmental nucleic acids — a feature leveraged for molecular diagnostics. For bacteria heterologously expressing the NbaSPARDA system, defense against invading bacteriophages and plasmids relies on filament formation. Collectively, these findings illustrate the detailed working mechanism of the NbaSPARDA complex and highlight the importance of its filament formation in host defense.
DOI: 10.1038/s41422-025-01100-z
Source: https://www.nature.com/articles/s41422-025-01100-z
Cell Research:《细胞研究》,创刊于1990年。隶属于施普林格·自然出版集团,最新IF:20.057
官方网址:https://www.nature.com/cr/
投稿链接:https://mts-cr.nature.com/cgi-bin/main.plex
