上海科技大学王皞鹏研究团队发现,配体诱导的泛素化通过阻碍信号基序的膜隔离释放LAG3免疫检查点功能。相关论文于2025年3月17日发表于国际顶尖学术期刊《细胞》杂志上。
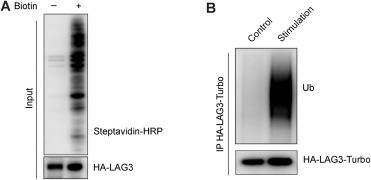
淋巴细胞活化基因3 (LAG3)已成为一个有前景的癌症免疫治疗靶点,但LAG3在配体结合后激活的机制尚不清楚。发现LAG3在配体结合后发生非K48连锁多泛素化,这促进了LAG3的抑制功能,而不是引导降解。这种泛素化可由主要组织相容性复合体II类(MHC II类)和膜结合(但不溶性)纤维蛋白原样蛋白1 (FGL1)的结合触发。LAG3泛素化由E3连接酶c-Cbl和cbl-b冗余介导,破坏了近膜基本残基序列的膜结合,从而稳定了LAG3细胞质尾部的膜解离构象,从而实现信号转导。
此外,LAG3泛素化对于LAG3介导的体内抗肿瘤免疫抑制至关重要。一致地,LAG3治疗性抗体抑制LAG3泛素化,与其检查点阻断作用相关。
此外,患者队列分析表明,LAG3/CBL共表达可以作为对LAG3阻断反应的生物标志物。总的来说,他们的研究揭示了在癌症免疫治疗中具有转化潜力的免疫检查点触发机制。
附:英文原文
Title: Ligand-induced ubiquitination unleashes LAG3 immune checkpoint function by hindering membrane sequestration of signaling motifs
Author: Yong Jiang, Anran Dai, Yuwei Huang, Hua Li, Jian Cui, Haochen Yang, Lu Si, Tao Jiao, Zhengxu Ren, Ziwei Zhang, Si Mou, Hengrui Zhu, Wenhui Guo, Qiang Huang, Yilin Li, Manman Xue, Jingwei Jiang, Fei Wang, Li Li, Qinying Zhong, Kun Wang, Baichuan Liu, Jinjiao Wang, Gaofeng Fan, Jun Guo, Liang Chen, Creg J. Workman, Zhirong Shen, Yan Kong, Dario A.A. Vignali, Chenqi Xu, Haopeng Wang
Issue&Volume: 2025-03-17
Abstract: Lymphocyte activation gene 3 (LAG3) has emerged as a promising cancer immunotherapy target, but the mechanism underlying LAG3 activation upon ligand engagement remains elusive. Here, LAG3 was found to undergo robust non-K48-linked polyubiquitination upon ligand engagement, which promotes LAG3’s inhibitory function instead of causing degradation. This ubiquitination could be triggered by the engagement of major histocompatibility complex class II (MHC class II) and membrane-bound (but not soluble) fibrinogen-like protein 1 (FGL1). LAG3 ubiquitination, mediated redundantly by the E3 ligases c-Cbl and Cbl-b, disrupted the membrane binding of the juxtamembrane basic residue-rich sequence, thereby stabilizing the LAG3 cytoplasmic tail in a membrane-dissociated conformation enabling signaling. Furthermore, LAG3 ubiquitination is crucial for the LAG3-mediated suppression of antitumor immunity in vivo. Consistently, LAG3 therapeutic antibodies repress LAG3 ubiquitination, correlating with their checkpoint blockade effects. Moreover, patient cohort analyses suggest that LAG3/CBL coexpression could serve as a biomarker for response to LAG3 blockade. Collectively, our study reveals an immune-checkpoint-triggering mechanism with translational potential in cancer immunotherapy.
DOI: 10.1016/j.cell.2025.02.014
Source: https://www.cell.com/cell/abstract/S0092-8674(25)00199-0
