CRISPR筛选和空间转录组学同时揭示了细胞内、细胞间和功能性转录回路,这一成果由麻省理工学院和哈佛大学的布罗德研究所Samouil L. Farhi研究组经过不懈努力而取得。该研究于2025年3月12日发表于国际一流学术期刊《细胞》杂志上。
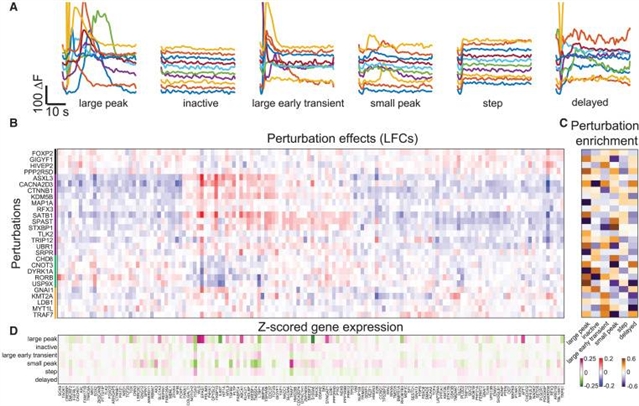
小组提出了成像空间转录组学与平行光学检测原位扩增引导RNA(Perturb-FISH)的结合。在培养单核细胞的脂多糖反应筛选中,Perturb-FISH恢复了与基于单细胞RNA测序的扰动效应(Perturb-seq)读数一致的细胞内效应,并揭示了先天免疫反应的细胞间和密度依赖性调节。同样,在三维异种移植物模型中,Perturb-FISH识别基因敲除改变的肿瘤免疫相互作用。当与人类诱导的多能干细胞(hIPSC)星形胶质细胞中自闭症谱系障碍风险基因的单独筛选中的功能读数配对时,Perturb-FISH显示出常见的钙活性表型及其相关的遗传相互作用和失调的分子途径。Perturb-FISH是在单细胞分辨率下研究空间和功能生物学的遗传和分子关联的一般方法。
据了解,集合光学屏幕已经使大规模的细胞相互作用、形态学或动力学研究成为可能,但它们还没有充分利用高度复杂的单细胞分解转录组读数的能力来告知分子途径。
附:英文原文
Title: Simultaneous CRISPR screening and spatial transcriptomics reveal intracellular, intercellular, and functional transcriptional circuits
Author: Loc Binan, Aiping Jiang, Serwah A. Danquah, Vera Valakh, Brooke Simonton, Jon Bezney, Robert T. Manguso, Kathleen B. Yates, Ralda Nehme, Brian Cleary, Samouil L. Farhi
Issue&Volume: 2025-03-12
Abstract: Pooled optical screens have enabled the study of cellular interactions, morphology, or dynamics at massive scale, but they have not yet leveraged the power of highly plexed single-cell resolved transcriptomic readouts to inform molecular pathways. Here, we present a combination of imaging spatial transcriptomics with parallel optical detection of in situ amplified guide RNAs (Perturb-FISH). Perturb-FISH recovers intracellular effects that are consistent with single-cell RNA-sequencing-based readouts of perturbation effects (Perturb-seq) in a screen of lipopolysaccharide response in cultured monocytes, and it uncovers intercellular and density-dependent regulation of the innate immune response. Similarly, in three-dimensional xenograft models, Perturb-FISH identifies tumor-immune interactions altered by genetic knockout. When paired with a functional readout in a separate screen of autism spectrum disorder risk genes in human-induced pluripotent stem cell (hIPSC) astrocytes, Perturb-FISH shows common calcium activity phenotypes and their associated genetic interactions and dysregulated molecular pathways. Perturb-FISH is thus a general method for studying the genetic and molecular associations of spatial and functional biology at single-cell resolution.
DOI: 10.1016/j.cell.2025.02.012
Source: https://www.cell.com/cell/abstract/S0092-8674(25)00197-7
