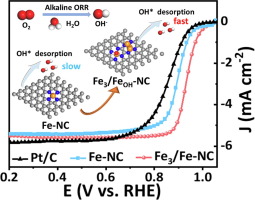
在本研究中,研究人员引入了一种采用均质少原子Fe3的修饰策略来调节Fe-N4的自旋极化。实验研究和理论计算表明,Fe(OH) -N4基序对OH配体的吸附明显增强,结构由方形平面场(Fe -N4)转变为方形金字塔场结构(Fe(OH) -N4)。这种结构转变降低了Fe-N4的3dxz、3dyz和3dz2轨道的自旋极化,导致三维轨道内未配对电子的减少。因此,这种调制导致反应中间体的吸附/解吸能适中,从而促进了ORR过程。
此外,原位光谱证实,与原子Fe-NC相比,Fe3/Fe(OH) -NC基序上OH*的脱附更有利,表明ORR的能垒更低。因此,Fe3/Fe-NC催化剂在0.1mol L−1 HClO4溶液中的半波电位为0.836 V,与可逆氢电极(RHE)相比,表现出优异的ORR性能。它还具有优异的锌空气电池效率。他们的研究引入了一种利用单原子与原子探针之间的相互作用来调节单原子催化剂电子结构的新方法。
据悉,Fe-N4基序被认为是氧还原反应(ORR)的主要非贵金属催化剂,具有取代铂(Pt)的潜力,但达到或超越铂基催化剂的性能仍然是一个重大挑战。
附:英文原文
Title: Spin polarization regulation of Fe–N4 by Fe3 atomic clusters for highly active oxygen reduction reaction
Author: Bin Wang a, Bingbao Mei b, Chao Liang a, Shengchun Yang a
Issue&Volume: 2025/03/10
Abstract: The Fe–N4 motif is regarded as a leading non-precious metal catalyst for the oxygen reduction reaction (ORR) with the potential to replace platinum (Pt), yet achieving or surpassing the performance of Pt-based catalysts remains a significant challenge. In this study, we introduce a modification strategy employing homogeneous few-atom Fe3 cluster to regulate the spin polarization of Fe–N4. Experimental research and theoretical calculations show that the incorporation of the Fe3 cluster significantly enhances the adsorption of Fe–N4 motif toward OH ligands, leading to a structural transformation from a square-planar field (Fe–N4) to a square-pyramid field structure (Fe(OH) –N4). This structural transformation reduces the spin polarization of 3dxz, 3dyz, and 3dz2 orbitals of Fe–N4, resulting in a decrease in unpaired electrons within 3d orbitals. As a result, this modulation leads to moderate adsorption/desorption energies of reaction intermediates, thereby facilitating the ORR process. Moreover, the in-situ spectroscopy confirms that the desorption of OH* on Fe3/Fe(OH) –NC motif is more favorable compared to atomic Fe–NC, indicating a lower energy barrier for ORR. Consequently, the Fe3/Fe–NC catalyst demonstrates outstanding ORR performance with a half-wave potential of 0.836 V vs. reversible hydrogen electrode (RHE) in 0.1 mol L-1 HClO4 solution and 0.936 V vs. RHE in 0.1 mol L-1 KOH solution, even surpassing commercial Pt/C catalyst. It also exhibits excellent Zn–air battery efficiency. Our study introduces a novel approach to modulating the electronic structure of single atoms catalysts by leveraging the robust interaction between single atoms and atomic clusters.
DOI: 10.1016/j.scib.2025.02.041
Source: https://www.sciencedirect.com/science/article/abs/pii/S2095927325002336
Science Bulletin:《科学通报》,创刊于1950年。隶属于SciEngine出版平台,最新IF:18.9
官方网址:https://www.sciengine.com/SB/home
投稿链接:https://mc03.manuscriptcentral.com/csb
