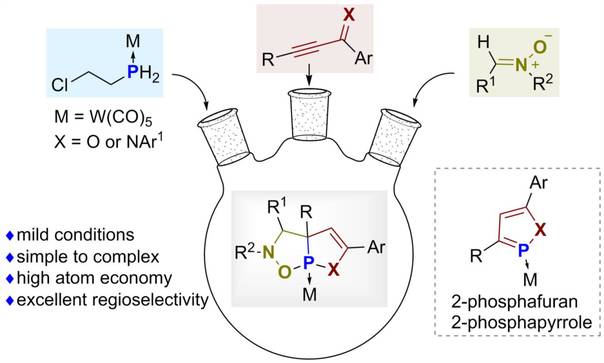
郑州大学段征小组提出了涉及对杂芳烃和硝基酮脱芳[3+2]环加成的异恶唑烷融合磷烯骨架串联路线。2025年2月19日,国际知名学术期刊《中国化学》发表了这一成果。
该课题组研究人员开发了一种串联方法,通过磷酸-迈克尔加成、分子内环化和脱芳化[3+2]环加成反应,将简单的β-氯乙基膦、炔基亚胺(或炔基酮)和硝基组装成结构复杂的异恶唑烷为主题的磷烯支架。异恶唑烷为主题的磷烯有三个杂原子,包括一个结磷原子。在去除钨基后,这些化合物可以作为潜在的p -立体配体,并可能具有生物活性。与吡咯和呋喃相反,芳香的2-磷吡咯和2-磷呋喃在脱芳[3+2]环加成反应中是很好的2π电子候选物,因为C=P部分的2p-3p轨道重叠较少。
据介绍,随着有机化学的发展,用简单的起始材料有效组装结构复杂的分子支架的需求不断增长。
附:英文原文
Title: A Tandem Route toward Isoxazolidine Fused Phospholene Skeleton Involving Dearomative [3+2] Cycloaddition of P-Heteroarenes and Nitrones
Author: Chenyong Xu, Yingqiang Wang, Meng Xiao, Rongqiang Tian, Zheng Duan
Issue&Volume: 2025-02-19
Abstract: The demands for effective assembly of structurally complex molecular scaffolds from simple starting materials are continuously growing along with the development of organic chemistry. We have developed a tandem approach that assembles simple β-chloroethylphosphane, alkynyl imines (or alkynyl ketones), and nitrones into structurally complex isoxazolidine fused phospholene scaffolds through a sequential process involving phospha-Michael addition, intramolecular cyclization, and dearomatizing [3+2] cycloaddition reactions. The isoxazolidine-fused phospholene has three heteroatoms, including a junction phosphorus atom. After removing the coordinated tungsten group, these compounds can serve as potential P-stereogenic ligands and may have biological activities. Contrary to pyrroles and furans, the aromatic 2-phosphapyrroles and 2-phosphafurans are good 2π-electron candidates in the dearomative [3+2] cycloaddition reactions due to the poor overlap of the 2p-3p orbitals of the C=P moiety.
DOI: 10.1002/cjoc.202401319
Source: https://onlinelibrary.wiley.com/doi/full/10.1002/cjoc.202401319
Chinese Journal of Chemistry:《中国化学》,创刊于1983年。隶属于Wiley,最新IF:5.4
官方网址:https://onlinelibrary.wiley.com/journal/16147065
投稿链接:https://mc.manuscriptcentral.com/cjoc
