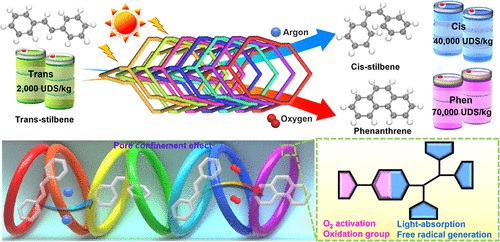
二苯乙烯的选择性光异构化或光环化以实现价值升级在工业应用中具有重要意义,但在温和条件下通过一锅光催化策略实现这两者仍然是一个挑战。
该文通过N,N,N-四(4-氨基苯基)-1,4-苯二胺(光吸收和自由基产生)和5,5′-(2,1,3-苯并噻二唑-4,7-二基)双[2-噻吩甲醛](催化中心)之间的共价偶联,合成了七重互穿三维共价有机框架(TPDT-COF)。由此获得的七重互穿结构提供了一种具有可调光催化能力和特定孔限制效应的功能性孔道,可用于选择性二苯乙烯的光异构化和光环化。
值得注意的是,通过在温和条件下简单地改变气体气氛(Ar、SeleCis>99%、SelePhen<1%和O2、SeleCiss<1%和SelePhen>99%),它能够以>99%的选择性光生顺式二苯乙烯或菲。理论计算证明,不同的气体环境对反应中间体的能垒有不同的影响,孔限制效应起着协同催化作用,从而诱导不同的产物生成。
该项研究可能有助于多孔晶体材料在选择性光异构化和光环化方面的探索。
附:英文原文
Title: Interpenetrating 3D Covalent Organic Framework for Selective Stilbene Photoisomerization and Photocyclization
Author: Tao-Yuan Yu, Qian Niu, Yifa Chen, Meng Lu, Mi Zhang, Jing-Wen Shi, Jiang Liu, Yong Yan, Shun-Li Li, Ya-Qian Lan
Issue&Volume: April 18, 2023
Abstract: The selective photoisomerization or photocyclization of stilbene to achieve value upgrade is of great significance in industry applications, yet it remains a challenge to accomplish both of them through a one-pot photocatalysis strategy under mild conditions. Here, a sevenfold interpenetrating 3D covalent organic framework (TPDT-COF) has been synthesized through covalent coupling between N,N,N,N-tetrakis(4-aminophenyl)-1,4-benzenediamine (light absorption and free radical generation) and 5,5′-(2,1,3-benzothiadiazole-4,7-diyl)bis[2-thiophenecarboxaldehyde] (catalytic center). The thus-obtained sevenfold interpenetrating structure presents a functional pore channel with a tunable photocatalytic ability and specific pore confinement effect that can be applied for selective stilbene photoisomerization and photocyclization. Noteworthily, it enables photogeneration of cis-stilbene or phenanthrene with >99% selectivity by simply changing the gas atmosphere under mild conditions (Ar, SeleCis. > 99%, SelePhen. < 1% and O2, SeleCis. < 1%, and SelePhen. > 99%). Theoretical calculations prove that different gas atmospheres possess varying influences on the energy barriers of reaction intermediates, and the pore confinement effect plays a synergistically catalytic role, thus inducing different product generation. This study might facilitate the exploration of porous crystalline materials in selective photoisomerization and photocyclization.
DOI: 10.1021/jacs.2c12313
Source: https://pubs.acs.org/doi/10.1021/jacs.2c12313
JACS:《美国化学会志》,创刊于1879年。隶属于美国化学会,最新IF:16.383
官方网址:https://pubs.acs.org/journal/jacsat
投稿链接:https://acsparagonplus.acs.org/psweb/loginForm?code=1000
