|
|
|
|
|
从2D到3D的癌细胞模型——纳米药物输送系统研究的谜团 | MDPI Nanomaterials |
|
|
论文标题:From 2D to 3D Cancer Cell Models—The Enigmas of Drug Delivery Research
期刊:Nanomaterials
作者:Indra Van Zundert, Beatrice Fortuni and Susana Rocha
发表时间:11 November 2020
DOI:10.3390/ nano10112236
微信链接:
https://mp.weixin.qq.com/s?__biz=MzI1MzEzNjgxMQ==&mid=2649978682&idx=4&sn=
e21f4cb033eaf9eb862766c24dbf0c64&chksm=f1decdfec6a944e83a18bd2662e319e404
1e5d597c19b2bf120969953a6bf736d6729892f865&token=940521072&lang=zh_CN#rd
期刊链接:https://www.mdpi.com/journal/nanomaterials
研究背景
迄今为止,癌症仍然是全球范围内导致死亡的主要原因之一。针对癌细胞的纳米药物载体目前是癌症治疗的有效手段之一,因而这种靶向治疗也受到了广泛关注。近年来,由于多功能纳米粒子的特性,使得其在生物医学应用领域取得了巨大研究进展,然而精心设计的纳米粒子治疗实验很少能达到临床阶段,因为虽然其在标准2D细胞培养系统中的结果较好,但往往在体内模型临床研究中的结果却不尽如人意 [1]。相比之下,3D细胞培养系统模型能够良好的模拟体内肿瘤生物学环境,因此受到越来越多的学者关注并激发他们进行了大量深入的研究。
来源:Heho Cancer
为了给用于纳米载体癌症治疗的3D癌症细胞模型研究人员提供全面指南,来自鲁汶大学(Catholic University of Louvain) 的Susana Rocha教授及其研究团队针对该研究在Nanomaterials期刊上发表了一篇综述型论文。在这篇综述中,作者概述了目前各种不同的3D癌细胞模型,并讨论它们在纳米医学领域的应用,同时对照指出了每种3D癌细胞模型生物应用的优缺点,详细解释了每种模型与肿瘤组织的体内生理联系,此外还总结了每种模型在评估纳米载体设计特定方面的潜在贡献,为3D癌细胞模型在纳米药物生物医学领域的开发、研究和应用提供理论指导。
研究内容
该文章对目前可用的3D癌细胞模型的模拟原理和应用进行了全面的概述,将不同的三维细胞模型分为无支架3D模型、嵌入式支架3D模型和微流体平台模型。
1. 无支架3D模型
目前最流行的无支架3D细胞模型之一是多细胞肿瘤球形 (MCTS) 模型 [2]。最常用的制备球状体的方法如图1所示。大量研究表明,在该模型下纳米载体的特性例如纳米颗粒尺寸、颗粒表面电荷、表面功能化都能影响其在实体肿瘤中渗透和扩散的能力。
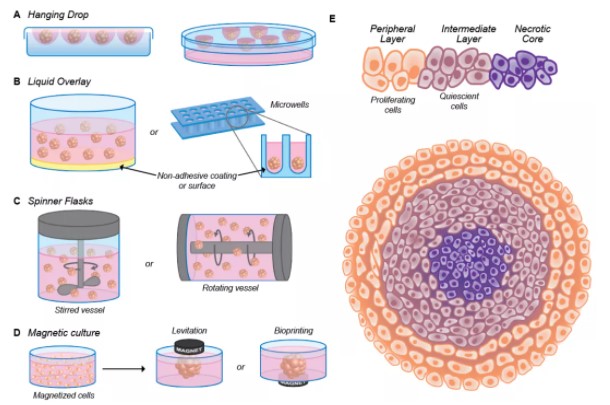
图1. 多细胞肿瘤球 (MCTS) 制备方法说明方案: (A) 悬滴法; (B) 液体叠加法; (C) 旋转瓶(搅拌或旋转容器)法; (D) 磁性培养法; (E) 描述增殖分布的球形生理学示意图。描绘了增殖细胞 (橙色,外层)、静止细胞 (紫色,中层) 和坏死细胞 (紫色,核心) 的分布。(注:细胞密度在外层较低)
2. 嵌入式支架3D模型
与无支架3D模型相比,使用嵌入式支架3D模型能够验证肿瘤细胞外围基质 (ECM) 模拟支架对纳米颗粒内化的影响。对于嵌入式支架3D模型,存在天然和合成两种不同类型的支架。最常用的天然支架是基于胶原蛋白、弹性蛋白、明胶、透明质酸聚合物基质或基质凝胶的支架,而合成类中聚乙醇酸 (PGA)、聚乳酸 (PLA) 和聚乙二醇 (PEG) 是最常用的支架材料。
3. 微流体平台模型
流体动力学是模拟体内生理至关重要的一面。微流控器件提供了一个可定制的平台来研究可控流动条件下的复杂 (多) 细胞结构。值得注意的是,在传统的2D细胞培养系统中没有液体流动会影响实验结果,这一点是3D细胞模型与之截然不同的地方。关于纳米颗粒在3D模拟癌症系统中的测试,研究人员开发了不同的微流控平台,如下图2所示。
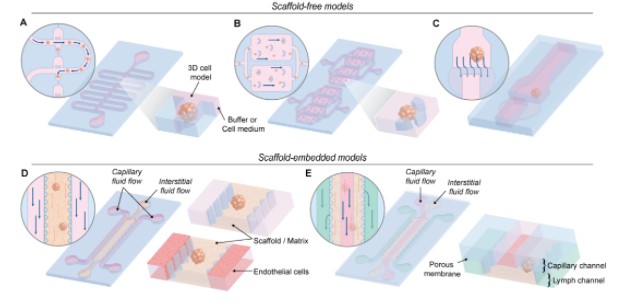
图2. 不同微流控装置的原理图。(A) 弯曲微通道装置; (B) 永久u形微结构的装置; (C) 双层微流控芯片装置; (D) 梯形柱体装置; (E) 三通道装置:毛细血管、间质和淋巴通道。
研究总结
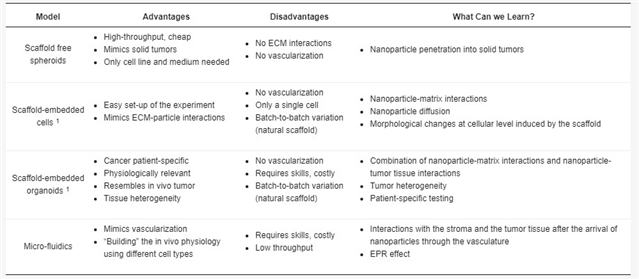
本文对目前可用的3D癌细胞模型的模拟原理和应用进行了全面的概述,将不同的三维细胞模型分为无支架模型、嵌入式支架模型和基于微流控的模型,并比较分析了三类模型的优缺点,以及各类模型目前所取得的成果各自在多大程度上影响了纳米医学的发展 (见图3)。
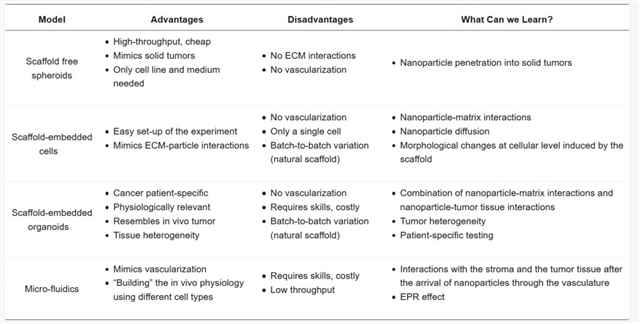
图3. 不同3D细胞培养系统模型系统各自优缺点以及它们为纳米医学发展提供的贡献比较说明。
在此基础上,作者对3D细胞培养系统模型在纳米医学上的应用机理以及表征手法等进行展望,强调了它们作为药物传递研究平台的潜力,并指出使用时的相关挑战,从而为3D细胞培养系统模型在纳米生物医学的应用奠定基础。此模型在体外和体内的研究之间搭建基础桥梁,最终为纳米颗粒转化到体内以及临床分期铺平道路。
参考文献
1. Bahrami,B.; Hojjat Farsangi,M.; Mohammadi,H.; Anvari,E.; Ghalamfarsa,G.; Youse?,M.; Jadidi-Niaragh,F. Nanoparticles and targeted drug delivery in cancer therapy. Immunol. Lett. 2017, 190, 64–83.
2. Lazzari, G.; Couvreur, P.; Mura, S. Multicellular tumor spheroids: A relevant 3D model for the in vitro preclinical investigation of polymer nanomedicines. Polym. Chem. 2017.
期刊简介
Nanomaterials (ISSN 2079-4991; IF 4.324)是一个与纳米材料研究相关的国际性开放获取期刊。目前已被SCIE、Scopus 等重要数据库收录。Nanomaterials 期刊采用单盲同行评审,一般审稿周期平均约为12.8天,稿件接收后约3.7天可见刊。
摘要:
Abstract
Over the past decades, research has made impressive breakthroughs towards drug delivery systems, resulting in a wide range of multifunctional engineered nanoparticles with biomedical applications such as cancer therapy. Despite these significant advances, well-designed nanoparticles rarely reach the clinical stage. Promising results obtained in standard 2D cell culture systems often turn into disappointing outcomes in in vivo models. Although the overall majority of in vitro nanoparticle research is still performed on 2D monolayer cultures, more and more researchers started acknowledging the importance of using 3D cell culture systems, as better models for mimicking the in vivo tumor physiology. In this review, we provide a comprehensive overview of the 3D cancer cell models currently available. We highlight their potential as a platform for drug delivery studies and pinpoint the challenges associated with their use. We discuss in which way each 3D model mimics the in vivo tumor physiology, how they can or have been used in nanomedicine research and to what extent the results obtained so far affect the progress of nanomedicine development. It is of note that the global scientific output associated with 3D models is limited, showing that the use of these systems in nanomedicine investigation is still highly challenging.
Research Content
1. Scaffold-Free 3D Model
One of the most popular 3D cell models currently used is the multicellular tumor spheroid (MCTS) model. Spheroids can be formed either via single cells that proliferate into cell aggregates or via pre-aggregated cell clusters that further proliferate. During cell proliferation, intercellular communication is established and the cells create their microenvironment. The most commonly used methods to prepare spheroids are illustrated in Figure 1.
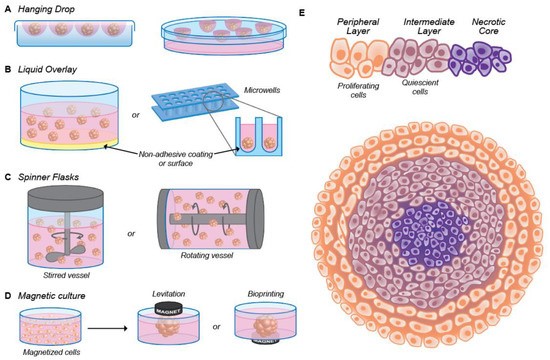
Figure 1. (A–D) Illustrative scheme of the methods for the preparation of multicellular tumor spheroid (MCTS):: the hanging drop method, where the cells come together at the bottom of a hanging drop (A), the liquid overlay method, where the surface of plates or (conical) wells is coated with a non-adhesive material (B), spinner flasks (stirred or rotating vessel), where the cells are kept in suspension (C) and magnetic culture, where, after the addition of magnetic particles, the cells are brought together through magnetic forces (D). (E) Schematic representation of the spheroid physiology depicting the distribution of proliferating (orange, outside layer), quiescent (violet, middle layer) and necrotic cells (purple, at the core). The cellular density is lower in the outside layer.
2.Scaffold-Embedded 3D Models
Although scaffold-free 3D models are suitable for studying the penetration of nanoparticles in solid tumors, the scaffold-free spheroid model lacks the so important tumor stroma, which the nanoparticles have to pass through in an in vivo setting. The tumor stroma consists of fibroblasts, endothelial cells, immune cells, and, importantly, ECM. Scaffold-embedded models can be used to verify the influence of the ECM mimicking scaffolds on nanoparticle internalization. In more detail, in this section, we will discuss single cells and miniature tumors embedded in a scaffold, which exhibit a respective lower and a higher level of complexity. It is important to mention that MCTS can also be embedded in a scaffold. However, as the specific advantages of the spheroid model were discussed in the previous section and the influence of the ECM is evaluated better in single embedded cells, embedded MCTSs will not be discussed.
2.3. Microfluidic Platforms
All the previously discussed models still lack one crucial aspect when it comes to mimicking the in vivo physiology: the fluid dynamics. Microfluidic devices offer a customizable platform to investigate complex (multi)cellular structures under controllable flow conditions. Importantly, the absence of fluid flow in conventional 2D cell culture systems can influence the results obtained.
In 3D cell models, particle sedimentation reduces the cell-particle contact time, which can hamper the use of these models to test the therapeutic efficiency of nanomedicines. To counteract this phenomenon, in recent years, microfluidic devices have attracted much attention, especially in drug testing. Concerning nanoparticle testing on 3D cancer-mimicking systems, different microfluidic platforms have been developed. As depicted in Figure 2, the microfluidic devices can be categorized into two groups: scaffold-free and scaffold-embedded models. In the second category, the flow might be applied directly on the scaffold or on an endothelial layer (or other cells) grown adjacent to the scaffold-embedded spheroid.
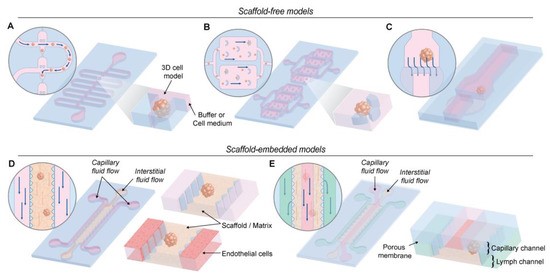
Figure 2. Schematic representation different microfluidic devices used. (A) A device containing a sinuous microchannel each of which contains five microwells for sedimentation trapping of the loaded microtissues or spheroids. (B) A device containing permanent U-shaped microstructures. The flow of culture medium towards the microstructure pushes the cells inside. (C) A two-layer microfluidic chip. The spheroid is immobilized in the higher part of the channel before the dam, where the height decreases from 250 to 25 µm. (D) These devices consist of 2 media channels engulfing an extended, central region containing the scaffold/matrix. The devices contain an array of trapezoidal posts that cage the matrix solution into well-defined regions with uniform surface interface. The side channels can be perfused with endothelial cells to simulate the endothelial wall. (E) Microfluidic platform with three types of channels - capillary, interstitial and lymphatic channels (noted with red, yellow and red colors). These channels are independently pressurized to mimic the elevated interstitial fluid pressure and are configured in a 3D structure by stacking two polydimethylsiloxane (PDMS) layers of microchannels with a porous membrane sandwiched between the layers.
Conclusions
In this review, we compared the 3D models currently available, pinpointed their pros and cons and described to what extent each model can contribute to nanomedicine development (Table 1). More specifically, nanoparticle accumulation and penetration in solid tumors can be investigated in depth using scaffold-free spheroid models. Using this 3D model, different research groups have shown that smaller particles can penetrate deeper in 3D multicellular structures. However, for improved circulation times, interactions with hepatocytes and renal clearance limit the nanoparticle size to 50 nm and 10 nm, respectively. This has led researchers to develop size-switching nanoparticles. Typically, these particles have an original diameter of 50–100 nm and, upon an external trigger, release smaller particles. Although this correlation between nanoparticle size and penetration depth is generally accepted, there are contradictory results from the influence of nanoparticle surface charge on spheroid penetration. Most reports suggest that negatively charged particles will penetrate further but it depends on the specific functional group exposed on the surface of the particle. In fact, functionalization with specific ligands or cell-penetrating peptides is also used to enhance the penetration of the particles in MCTS.
Table 1. Overview of 3D model systems, their respective advantages and disadvantages and what knowledge they can provide in nanoparticle research.
In addition to the technical challenges, there are also biological and physical issues are not yet fully understood. Especially nanoparticle and ECM interactions are still a “tough nut to crack.” The diffusion of nanoparticles through the scaffold can be improved by tuning their size and charge, however, there is only a handful of reports addressing this optimization step. Other overlooked aspects included the effect of the scaffold in the cell biology (receptor distribution, cytoskeleton organization) and the cellular heterogeneity. Implementation of the 3D culture systems can shed light into the reasons behind the different efficiency of nanomedicine in in vitro and in vivo models. More important, 3D models can build a fundamental bridge between in vitro and in vivo studies, ultimately paving the way for nanoparticle translation to in vivo and clinical stages.


(来源:科学网)
特别声明:本文转载仅仅是出于传播信息的需要,并不意味着代表本网站观点或证实其内容的真实性;如其他媒体、网站或个人从本网站转载使用,须保留本网站注明的“来源”,并自负版权等法律责任;作者如果不希望被转载或者联系转载稿费等事宜,请与我们接洽。